Molecular Genetic Analysis of Muscle Gene and Protein Function During Drosophila Development
We use the fruit fly, Drosophila melanogaster, to explore gene expression and protein function in muscle cells. Defects in contractile proteins cause human cardiac and skeletal muscle disease. Therefore, it is useful to pursue an understanding of contractile protein function in an organism that is readily manipulated by genetic and transgenic means. Our integrative approach allows us to study muscle from the molecular level (using biochemical and biophysical assays), through the cellular level (using microscopy and fiber mechanical assays) to the level of whole organism muscle function.
The molecular motor of muscle is myosin heavy chain, a protein that interacts with actin to cause muscle contraction. We determined that Drosophila possesses a single muscle myosin heavy chain gene, and that multiple forms of the myosin protein are produced by the process of alternative RNA splicing. Specific regions of the myosin RNA are included in specific muscle types to yield myosin protein isoforms. The regions that differ among the isoforms must be critical for the myosin-mediated ultrastructural and physiological differences among muscle types. In collaboration with Dr. Ron Milligan (the Scripps Research Institute), we determined the location of the isoform differences on the three dimensional map of the myosin molecule. Using these data, we developed hypotheses regarding how these differences can affect myosin function.
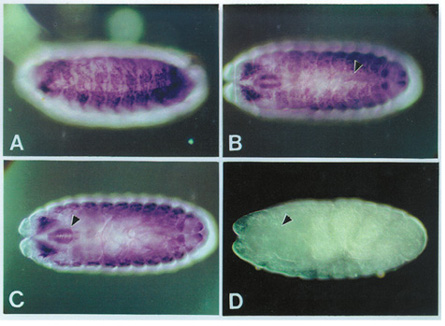 Tissue-specific accumulation
of alternative forms of myosin heavy chain RNA in Drosophila embryos (from
Zhang and Bernstein, 2001)
Tissue-specific accumulation
of alternative forms of myosin heavy chain RNA in Drosophila embryos (from
Zhang and Bernstein, 2001)

Stereo image of the myosin head with
locations of domains encoded by alternative exons highlighted (from Kronert
et al., 1999)
To experimentally test our hypotheses on myosin isoform function, we developed transgenic strains of Drosophila that express the wrong isoforms in particular muscle types. As an initial approach, we showed that the normal myosin gene can rescue muscle defects associated with myosin null mutations. Interestingly, additional copies of the myosin gene result in defective muscles as well, due to an overabundance of myosin-containing thick filaments. We next succeeded in expressing the embryonic form of myosin in adult flight muscle. To our surprise, the adult muscle assembles normally using the embryonic protein, indicating that structural properties of the muscle are not affected by the isoform of myosin present. However, the flight muscles do not function when the embryonic myosin substitutes for the normal form, indicating that myosin isoforms are functionally different.
We have produced additional transgenic fly lines that express other myosin isoforms, particular mutant myosins and putative suppressor mutations. Currently, we are isolating single isoforms of the myosin protein from these lines to determine the biochemical and biophysical properties that are encoded by particular variable regions of the protein, e.g. actin binding ability, ATPase kinetics, in vitro thin filament motility. Transient kinetic studies are performed in collaboration with Dr. Michael Geeves (University of Kent at Canterbury). Muscles with altered myosin isoforms are studied in collaboration with Dr. Douglas Swank (Rensselaer Polytechnique Institute) to assess how mechanical properties are affected by the alterations. Overall our approach should lead to a more complete understanding of the in vitro and in vivo properties imparted by particular regions of the myosin molecule.
We are dissecting the role of myosin domains and residues in Drosophila cardiac muscle in collaboration with Dr. Rolf Bodmer, Dr. Karen Ocorr (Sanford-Burnham Institute) and Dr. Anthony Cammarato (Johns Hopkins University). Using their quantitative videomicroscopy set-up, we have documented myosin-based defects that mimic human dilated cardiomyopathy and restrictive cardiomyopathy. We are also examining the effects of myosin mutations known to cause cardiomyopathy in humans, with the goal of developing approaches to suppress these defects by genetic or pharmacological treatments.
We are producing and analyzing Drosophila models of several myosin-based human diseases: inclusion body myopathy type 3, myosin storage myopathy and Freeman-Sheldon syndrome. The goal is to determine how the mutations affect muscle development and aging using an organism with a defined genetic background. Studies on muscle ultrastructure, myosin biochemistry and muscle physiology will aid in understanding the cause and effects of each disease.
We are also studying Drosophila UNC-45, a molecular chaperone/co-chaperone that aids in folding of muscle myosin heavy chain and possibly other muscle or non-muscle proteins. We solved the structure of the Drosophila UNC-45 protein in collaboration with Dr. Tom Huxford (SDSU). This will aid us in pursuing a detailed structure/function analysis of this protein in vitro and in vivo.
Our research on UNC-45 and myosin mutations will lead to insights as to how mutations that result in production of abnormally folded contractile proteins cause phenotypic defects and how these may be ameliorated. Since several neuromuscular diseases arise from aberrant protein folding and accumulation of misfolded protein aggregates, our work will contribute to understanding the disease process and may yield insight into genetic and pharmacological approaches that prove to be therapeutic in humans.
Recent Publications
Note: documents in Portable Document Format (PDF) require Adobe Acrobat Reader 5.0 or higher to view, download Adobe Acrobat Reader.
Miller, M. S., C. M. Dambacher, A. F. Knowles, J. M. Braddock, G P. Farman, T. C. Irving, D. M. Swank, S. I. Bernstein and D. W. Maughan (2009) Alternative S2 hinge regions of the myosin rod affect myofibrillar structure and myosin kinetics. Biophys. J. 96:4132-4143.
Bloemink, M. J., C. M. Dambacher, A. F. Knowles, G. C. Melkani, M. A. Geeves and S. I. Bernstein (2009) Alternative exon 9-encoded relay domains affect more than one communication pathway in the Drosophila myosin head. J. Mol. Biol. 389:707-721.
Ocorr, K., M. Fink, A. Cammarato, S. Bernstein and R. Bodmer. (2009) Semi-automated optical heartbeat analysis of small hearts. J. Vis. Exp. 31: 1435.
Kronert, W. A., G. C. Melkani, A. Melkani and S. I. Bernstein (2010) Mutating the converter-relay interface of Drosophila myosin perturbs ATPase activity, actin motility, myofibril stability and flight ability. J. Mol. Biol. 398:625-632.
Melkani, G. C., C. F. Lee, A. Cammarato and S. I. Bernstein (2010) Drosophila UNC-45 prevents heat-induced aggregation of skeletal muscle myosin and facilitates refolding of citrate synthase. Biochem. Biophys. Res. Comm. 396: 317-322.
Lee, C. F., G. C. Melkani, Q. Yu, J. A. Suggs, W. A. Kronert, Y. Suzuki, L. Hipolito, M. G. Price, H. F. Epstein and S. I. Bernstein (2011) Drosophila UNC-45 accumulates in embryonic blastoderm and in muscles and is essential for muscle myosin stability. J. Cell Sci. 24: 699-705.
Lee, C. F., A. V. Hauenstein, J. K. Fleming, W. C. Gasper, V. Engelke, B. Sankaran, S. I. Bernstein and T. Huxford (2011) X-ray crystal structure of the UCS domain-containing UNC-45 myosin chaperone from Drosophila melanogaster. Structure 19: 397-408.
Cammarato, A., C. H. Ahrens, N. N. Alayari, E. Qeli, J. Rucker, M. C. Reedy, C. M. Zmasek, M. Gucek, R. N. Cole, J. E. Van Eyk, R. Bodmer, B. O’Rourke, S. I. Bernstein and D. B. Foster (2011) A mighty small heart: the cardiac proteome of adult Drosophila melanogaster. PLoS One 6: e18497.
Melkani, G. C., R. Bodmer, K. Ocorr and S.I. Bernstein (2011) The UNC-45 chaperone is critical for establishing myosin-based myofibrillar organization and cardiac contractility in the Drosophila heart model. PLoS One 6: e22579.
Bloemink, M. J., G. C. Melkani, C. M. Dambacher, S. I. Bernstein and M. A. Geeves (2011) Two Drosophila myosin transducer mutants with distinct cardiomyopathies have divergent ADP and actin affinities. J. Biol. Chem.286: 28435–28443.
Ramanath, S., Q. Wang, S. I. Bernstein and D. M. Swank (2011) Disrupting the myosin converter-relay interface impairs Drosophila indirect flight muscle performance. Biophys. J.10: 1114-1122.
Cammarato, A., X. Li, M. C. Reedy, C. F. Lee, W. Lehman and S. I. Bernstein (2011) Structural basis for myopathic defects engendered by alterations in the myosin rod. J. Mol. Biol. 414: 477-484.
Kronert, W. A., G. C. Melkani, A. Melkani and S. I. Bernstein (2012) Alternative relay and converter domains tune native muscle myosin isoform function in Drosophila. J. Mol. Biol. 416: 543–557.
Kaushik, G., A. C. Zambon, A. Fuhrmann, S. I. Bernstein, R. Bodmer, A. J. Engler and A. Cammarato (2012) Measuring passive myocardial stiffness in Drosophila melanogaster to investigate diastolic dysfunction. J. Cell. Mol. Med. 16:1656-1662.
Caldwell, J. T., G. C. Melkani, T. Huxford and S. I. Bernstein (2012) Transgenic expression and purification of myosin isoforms using the Drosophila melanogaster indirect flight muscle system. Methods. 56: 25-32.
Wang. Y., G. C. Melkani, J. A. Suggs, A. Melkani, W. A. Kronert, A. Cammarato and S. I. Bernstein (2012) Expression of the inclusion body myopathy 3 mutation in Drosophila depresses myosin function and stability and recapitulates muscle inclusions and weakness. Mol. Biol. Cell. 23: 2057-2065.
Xie , H. B., A. Cammarato, N. S. Rajasekaran, H. Zhang, J. A. Suggs, H.-C. Lin, S. I. Bernstein, I. J. Benjamin and K. G. Golic (2013) The NADPH metabolic network regulates human alphaB crystallin cardiomyopathy and reductive stress in Drosophila melanogaster. PLoS Genet. 9: e1003544.
Melkani, G. C., A. S. Trujillo, R. Ramos, R. Bodmer, S. I. Bernstein and K. Ocorr (2013) Huntington's disease induced cardiac amyloidosis is reversed by modulating protein folding and oxidative stress pathways in the Drosophila heart. PLoS Genet. 9: e1004024.
Kronert, W.A., G. C. Melkani, A. Melkani and S. I. Bernstein (2014) Mapping interactions between myosin relay and converter domains that power muscle function. J. Biol. Chem. 289:12779-12790.
|